Tag: infectious disease modeling
Filter by:
The Coronavirus Outbreak: Part One – Modeling “S...

Since 2017, in modeling the threat from wildfire on communities in California, the significant new RMS innovation has been in capturing the…
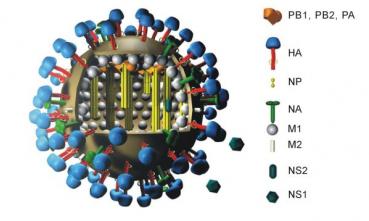
A Coup Against Flu?

Recent articles from two separate research groups published in Science and Nature Medicine report major breakthroughs in flu vaccine research…
Ebola in the U.S. – How big of problem are we lo...

As mentioned in my previous blog post, Ebola has the potential to be one of the deadliest epidemics in a century, but the primary area of…